Sulfur compounds in different proportions are contained in natural gas extracted from the fields for delivery to the consumer via pipelines. If you do not get rid of them, aggressive substances will destroy the pipeline, render the fittings unusable. In addition, when contaminated blue fuel is burned, toxins are released.
In order to avoid negative consequences, amine gas purification from hydrogen sulfide is carried out. This is the easiest and most inexpensive way to separate harmful components from a combustible mineral. We will tell you how the process of separation of sulfur inclusions proceeds, how the purification unit is arranged and works.
The content of the article:
- Purpose of cleaning fossil fuels
- Existing methods for separating hydrogen sulfide
- How a Typical Installation Works
- Four cleaning options with alconolamines
- Conclusions and useful video on the topic
Purpose of cleaning fossil fuels
Gas is the most popular fuel. It attracts with the most affordable price and causing the least damage to the environmental situation. The indisputable advantages include the simplicity of control of the combustion process and the ability to secure all stages of fuel processing in the course of obtaining thermal energy.
However, natural gaseous fossil is not mined in its pure form, because simultaneously with gas extraction from the well, associated organic compounds are pumped out. The most common of them is hydrogen sulfide, the content of which varies from tenths to ten percent or more, depending on the field.
Image gallery
Photo from
Natural gas is the most widespread and widely demanded type of fuel, the popularity of which is based not only on the affordability of the price
Most of the cooking stoves and cookers in the food industry run on main gas
The best option for heating large-scale industrial enterprises is gas. It does the least harm to the natural environment, does not emit soot and insoluble combustion products
Gas boilers are most often used in the preparation of hot water and heating of private houses / apartments, small and medium-sized commercial facilities, workshops.
Gas is used to obtain the required temperature of the working environment in the chemical and food industries
Natural gas is necessary to obtain industrial gases, which are then used in welding, in the power supply of various heaters
Main gas is used as a valuable raw material for the production of many chemical compounds, from which all kinds of polymer products are then made
Regardless of the purpose of using natural gas, it must be cleaned of hydrogen sulfide and other organic compounds before being delivered to the pipeline.
Natural gas is the most common fuel
Using gas in cooking
The use of gas in the heating of industrial enterprises
Atmospheric Burner Gas Boiler
The use of gas in industrial processes
Industrial gases production
The use of gas as a raw material in the chemical industry
Gas transportation through the gas pipeline
Hydrogen sulfide is poisonous, hazardous to the environment, and harmful to catalysts used in gas processing. As we have already noted, this organic compound is extremely aggressive towards steel pipes and metal valves.
Naturally, corroding the private system and main gas pipeline, hydrogen sulfide leads to leaks of blue fuel and related to this fact extremely negative, risky situations. To protect the consumer, compounds harmful to health are removed from the composition of the gaseous fuel even before it is delivered to the pipeline.
According to the standards of hydrogen sulfide compounds in the gas transported through the pipes, it cannot exceed 0.02 g / m³. However, in fact, there are much more of them. In order to achieve the value regulated by GOST 5542-2014, cleaning is required.
Existing methods for separating hydrogen sulfide
In addition to the hydrogen sulfide prevailing against the background of other impurities, blue fuel may contain other harmful compounds. You can find in it carbon dioxide, light mercaptans and carbon sulfide. But directly hydrogen sulfide will always prevail.
Image gallery
Photo from
The presence of organic impurities in natural gas is the main cause of corrosion of steel pipelines and fittings. Its results are deplorable
Due to the appearance of rust, the walls of the gas pipe become thinner. As a result, tightness is lost. At best, gas leaks will incur costs, at worst - explosions and poisoning
The rust that appears in the pipeline will quickly spread to the shut-off valves. Rusty taps and valves cannot be closed in a hazardous situation or for repair
Due to rust, a relief will appear inside the pipes, even a partial overlap of the route may form. The result of the aforementioned negative can be an explosion, one of the reasons for which is often the instability of pressure in the gas system.
Corrosion inside the gas pipe
Loss of tightness of the gas pipeline
Rusting of the steel fittings of the gas pipeline
Gas explosion due to unstable pressure
It should be noted that some insignificant content of sulfur compounds in the purified gaseous fuel is acceptable. The specific figure of the tolerance depends on the purposes for which the gas is produced. For example, for the production of ethylene oxide, the total sulfur content must be less than 0.0001 mg / m³.
The cleaning method is chosen based on the required result.
All currently existing methods are divided into two groups:
- Sorption. They consist in the absorption of hydrogen sulfide compounds by a solid (adsorption) or liquid (absorption) reagent, followed by the release of sulfur or its derivatives. After that, the harmful impurities released from the gas are disposed of or recycled.
- Catalytic. They consist in the oxidation or reduction of hydrogen sulfide with its transformation into elemental sulfur. The process is carried out in the presence of catalysts - substances that stimulate the course of a chemical reaction.
Adsorption involves the collection of hydrogen sulfide by concentrating it on the surface of a solid. Most often, granular materials based on activated carbon or iron oxide are used in the adsorption process. The large surface area characteristic of the grains maximizes the retention of sulfur molecules.

All blue fuel purification methods are subdivided into sorption and catalytic. Cleaning equipment is focused on the principle of operation of a particular technology. However, there are installations in which several methods are combined, due to which complex cleaning is carried out.
The absorption technology differs in that gaseous hydrogen sulfide impurities are dissolved in an active liquid substance. As a result, gaseous contaminants pass into the liquid phase. Then the isolated harmful components are removed by stripping, otherwise desorption, in this way they are removed from the reactive liquid.
Despite the fact that the adsorption technology refers to "dry processes" and allows you to produce fine purification of blue fuel, in the removal of contaminants from natural gas is more often used absorption. Collection and elimination of hydrogen sulfide compounds using liquid absorbers is more profitable and expedient.

The most popular type of adsorber is activated carbon, used in the form of capsules or grains. The surface of each element "absorbs" hydrogen sulfide and other organic inclusions
The absorption methods used in gas purification are divided into the following three groups:
- Chemical. They are produced using solvents that freely react with acidic hydrogen sulphide contaminants. Ethanolamines or alkanolamines have the highest absorption capacity among chemical sorbents.
- Physical. They are performed by physically dissolving gaseous hydrogen sulfide in a liquid absorber. Moreover, the higher the partial pressure of the gaseous pollutant, the faster the dissolution process proceeds. Methanol, propylene carbonate, etc. are used as an absorber.
- Combined. In the mixed version of hydrogen sulfide extraction, both technologies are involved. The main work is performed by absorption, and fine additional treatment is performed by adsorbents.
For half a century, the most demanded and popular technology for the extraction and removal from fossil fuels hydrogen sulfide and carbonic acid is a chemical gas purification using an amine sorbent used in the form of an aqueous solution.

Sorption methods of natural fuel purification are based on the ability of solid and liquid substances react with hydrogen sulfide and other organic impurities, thereby releasing them from the composition gas
Amine technology is more suitable for handling large volumes of gas because:
- Lack of deficit. Reagents can always be purchased in the amount required for purification.
- Acceptable absorbency. Amines are highly absorbent. Of all the substances used, only they are able to remove 99.9% of hydrogen sulfide from the gas.
- Priority characteristics. Aqueous amine solutions are distinguished by the maximum acceptable viscosity, vapor density, thermal and chemical stability, and low heat capacity. Their characteristics ensure the best possible absorption process.
- No toxicity of reactive substances. This is an important argument that convinces people to resort to the amine method.
- Selectivity. Quality required for selective absorption. It allows the necessary reactions to be carried out sequentially in the order required for optimal results.
Ethanolamines used in chemical methods of gas purification from hydrogen sulfide and carbon dioxide include monoethanolamines (MEA), diethanolamines (DEA), triethanolamines (TEA). Moreover, substances with prefixes mono- and di- are removed from the gas and H2S, and CO2. But the third option helps to remove only hydrogen sulfide.
When performing selective cleaning of blue fuel, methyldiethanolamines (MDEA), diglycolamines (DHA), diisopropanolamines (DIPA) are used. Selective absorbents are mainly used abroad.
Naturally ideal absorbents to meet all cleaning requirements prior to delivery to the system gas heating and the supply of other equipment does not yet exist. Each solvent has some pluses along with minuses. When choosing a reactive substance, they simply determine the most suitable from a number of proposed ones.
How a Typical Installation Works
Maximum absorption capacity in relation to H2S is characterized by a solution of monoethanolamine. However, this reagent has a couple of significant disadvantages. It is distinguished by a rather high pressure and the ability, during the operation of the amine gas purification unit, to create irreversible compounds with carbon sulfide.
The first disadvantage is eliminated by flushing, as a result of which the amine vapor is partially absorbed. The second is rarely encountered during the processing of field gases.
Image gallery
Photo from
Extraction of hydrogen sulfide and associated organic components from natural fossil fuels is carried out at absorption plants
Installations can be constructed near the field, installed on the route or in front of the entrance to the gas processing plant. In any case, cleaning is carried out before supplying gaseous fuel to the consumer.
Gas purification measures and equipment used are constantly being improved. If earlier the sulfur separated from the composition of the natural gaseous mixture was simply utilized, now it are stored and sent to the production of sulfuric acid, paper, carbon dioxide, dry ice, rubber and more another
Absorber cleaning is not cheap. It significantly increases the cost of the processed fuel. However, the multiple use of the amine solution in the installation allows you to reduce the cost.
Absorption plant for the extraction of hydrogen sulfide from gas
A complex of treatment plants on the highway
Advanced gas treatment complexes
Natural gas treatment plant pipeline
The concentration of an aqueous solution of monoethanolamine is selected empirically, based on the studies carried out, it is taken to purify gas from a certain field. The selection of the percentage of the reagent takes into account its ability to withstand the aggressive effects of hydrogen sulfide on the metal components of the system.
Typical absorbent content is usually in the range from 15 to 20%. However, it often happens that the concentration is increased to 30% or decreased to 10%, depending on how high the degree of purification should be. Those. for what purpose, in heating or in the production of polymer compounds, gas will be used.
Note that an increase in the concentration of amine compounds decreases the corrosive potential of hydrogen sulfide. But it must be taken into account that in this case the consumption of the reagent increases. Consequently, the cost of the treated commercial gas rises.
The main unit of the purification plant is the absorber of the disc or packaged version. This is a vertically oriented apparatus resembling a test tube, with nozzles or plates located inside. In the lower part of it there is an inlet for the supply of the raw gas mixture, in the upper part there is an outlet to the scrubber.

If the gas to be cleaned in the installation is under pressure sufficient for the passage of the reagent to the heat exchanger and then to the stripping column, the process takes place without the participation of a pump. If the pressure is not enough for the flow of the process, the outflow is stimulated by pumping technology.
The gas stream, after passing through the inlet separator, is injected into the lower section of the absorber. It then passes through trays or nozzles located in the middle of the housing, on which contaminants settle. The nozzles, completely moistened with amine solution, are separated by grids for even distribution of the reagent.
Then the blue fuel cleaned from contamination is sent to the scrubber. This device can be connected in the recycling circuit after the absorber or located in the upper part of it.
The spent solution flows down the walls of the absorber and is sent to the stripping column - a desorber with a boiler. There the solution is purified from the absorbed contaminants by the vapors released during boiling water in order to return back to the installation.
Regenerated, i.e. freed from hydrogen sulfide compounds, the solution flows into the heat exchanger. In it, the liquid is cooled in the process of transferring heat to the next portion of the contaminated solution, after which it is pumped into the refrigerator for complete cooling and condensation of steam.
The cooled absorbent solution is fed back to the absorber. This is how the reagent circulates through the installation. Its vapors are also cooled and purified from acidic impurities, after which they replenish the reagent supply.
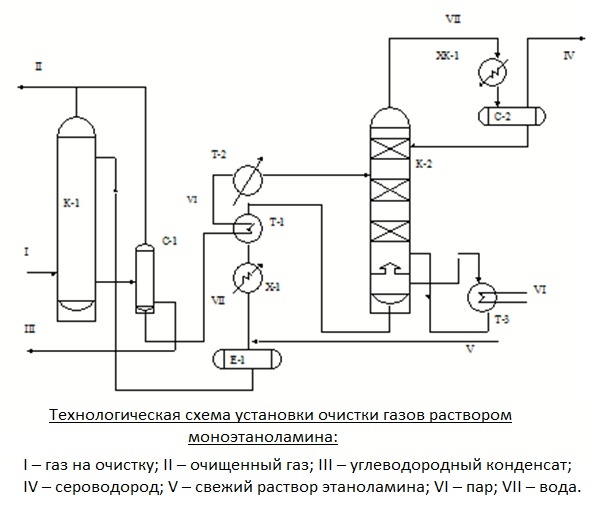
Most often, gas purification schemes are used with monoethanolamine and diethanolamine. These reagents make it possible to extract not only hydrogen sulfide, but also carbon dioxide from the blue fuel.
If it is necessary to simultaneously remove CO from the treated gas2 and H2S, two-stage cleaning is performed. It consists in the use of two solutions, differing in concentration. This option is more economical than one-step cleaning.
First, gaseous fuel is cleaned with a strong composition with a reagent content of 25-35%. Then the gas is treated with a weak aqueous solution, in which the active substance is only 5-12%. As a result, both coarse and fine cleaning is carried out with a minimum consumption of solution and a reasonable use of the generated heat.
Four cleaning options with alconolamines
Alkonolamines or amino alcohols are substances containing not only an amine group, but also a hydroxy group.
The design of installations and technologies for the purification of natural gas with alkanolamines differ mainly in the method of supplying the absorbing substance. There are four main methods most often used in gas cleaning using this type of amine.
The first way. It predetermines the supply of the active solution in one stream from the top. The entire volume of absorbent is directed to the upper tray of the installation. The cleaning process takes place at a temperature background not higher than 40 ° C.

The simplest cleaning method involves feeding the active solution in one stream. This technique is used if there is a small amount of impurities in the gas.
This technique is usually used for light contamination with hydrogen sulphide compounds and carbon dioxide. In this case, the total thermal effect for the production of commercial gas is, as a rule, low.
Second way. This treatment option is used when there is a high content of hydrogen sulfide compounds in gaseous fuels.
In this case, the reactive solution is fed into two streams. The first, with a volume of about 65-75% of the total mass, is sent to the middle of the installation, the second is supplied from above.
The amine solution flows down the trays and meets the ascending gas streams that are pumped into the lower tray of the absorption unit. Before serving, the solution is heated to no more than 40 ° C, but during the interaction of the gas with amine, the temperature rises significantly.
To prevent the cleaning efficiency from dropping due to an increase in temperature, excess heat is removed along with the waste solution saturated with hydrogen sulfide. And at the top of the unit, the flow is cooled in order to extract the residues of acidic components together with condensate.
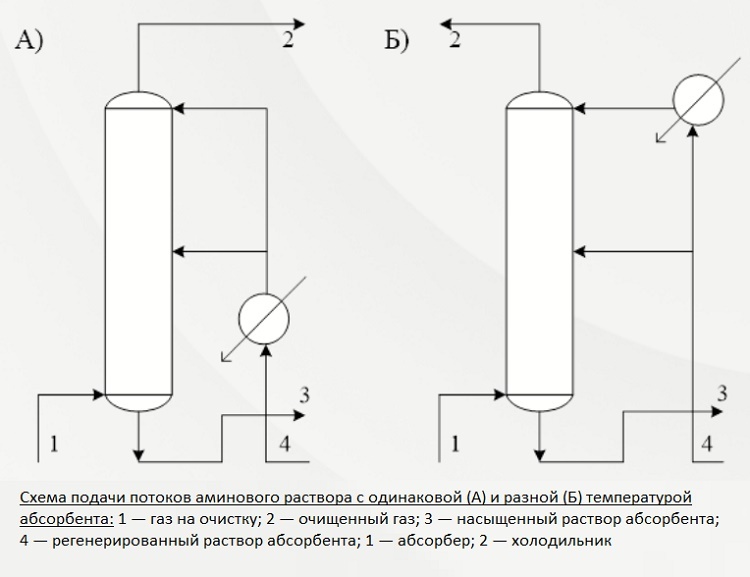
The second and third of the described methods predetermine the supply of the absorption solution in two streams. In the first case, the reagent is supplied at the same temperature, in the second - at different
It is an economical way to reduce the consumption of both energy and active solution. Additional heating is not performed at any stage. Technologically, it is a two-level purification, which makes it possible to prepare commercial gas for supply to the main line with the least amount of losses.
Third way. It involves the delivery of the absorber to the cleaning unit in two streams of different temperatures. The technique is used if, in addition to hydrogen sulfide and carbon dioxide, there is also CS in the raw gas2, and COS.
The predominant part of the absorber, approximately 70-75%, heats up to 60-70 ° C, and the remaining part only up to 40 ° C. The streams are supplied to the absorber in the same way as in the above-described case: from the top and into the middle.
The formation of a zone with a high temperature makes it possible to quickly and efficiently remove organic contaminants from the gas mass at the bottom of the cleaning column. And at the top, carbon dioxide and hydrogen sulfide are precipitated by an amine of standard temperature.
Fourth way. This technology predetermines the supply of an aqueous amine solution in two streams with different degrees of regeneration. That is, one is supplied unrefined, containing hydrogen sulfide inclusions, the second without them.
The first stream cannot be called completely polluted. It only partially contains acidic components, because some of them are removed during cooling to + 50 ° / + 60 ° C in the heat exchanger. This solution stream is taken from the bottom of the stripper, cooled and directed to the middle of the column.
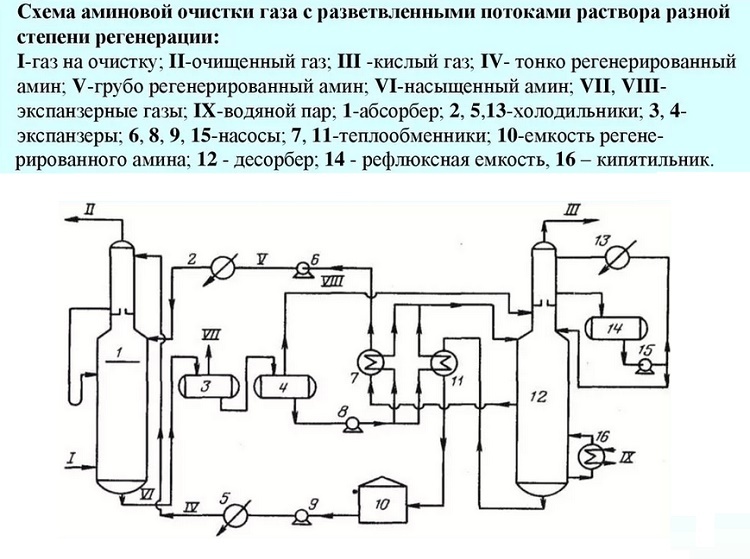
With a significant content of hydrogen sulfide and carbon dioxide components in gaseous fuel, cleaning is carried out with two streams of solution with different degrees of regeneration
Only that part of the solution, which is pumped into the upper sector of the installation, undergoes deep cleaning. The temperature of this stream usually does not exceed 50 ° C. This is where the fine cleaning of gaseous fuels is carried out. This scheme allows you to reduce costs by at least 10% by reducing steam consumption.
It is clear that the cleaning method is chosen based on the presence of organic contaminants and economic feasibility. In any case, the variety of technologies allows you to choose the best option. On the same amine gas treatment unit, the degree of purification can be varied, obtaining a blue fuel with the necessary gas boilers, stoves, heaters characteristics.
Conclusions and useful video on the topic
The following video will acquaint you with the specifics of extracting hydrogen sulfide from associated gas produced together with oil by an oil well:
The installation for the purification of blue fuel from hydrogen sulfide with the production of elemental sulfur for further processing will be presented by the video:
The author of this video will tell you how to get rid of hydrogen sulfide from biogas at home:
The choice of a gas purification method is primarily focused on solving a specific problem. The artist has two ways: to follow a proven scheme, or to prefer something new. However, the main guideline should still be economic feasibility while maintaining quality and obtaining the desired degree of processing.


